(C) 2013 Sandra Knapp. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
Citation: Knapp S, Vorontsova MS (2013) From introduced American weed to Cape Verde Islands endemic: the case of Solanum rigidum Lam. (Solanaceae, Solanum subgenus Leptostemonum). PhytoKeys 25: 35–46. doi: 10.3897/phytokeys.25.4692
A Solanum species long considered an American introduction to the Cape Verde Islands off the west coast of Africa is identified as Solanum rigidum, a member of the Eggplant clade of Old World spiny solanums (Solanum subgenus Leptostemonum) and is probably endemic to the Cape Verde Islands. Collections of this species from the Caribbean are likely to have been introduced from the Cape Verde Islands on slave ships. We discuss the complex nomenclatural history of this plant and provide a detailed description, illustration and distribution map. The preliminary conservation status of Solanum rigidum is Least Concern, but needs to be reassessed in light of its endemic rather than introduced status.
Africa, aubergine, Cape Verde Islands, Caribbean, description, eggplant, typification
Solanum L. (Solanaceae) is the largest genus of Solanaceae; with some 1400 species, it is one of the largest angiosperm genera (
Although Solanum is predominantly a New World group, two clades of Old World species are resolved in molecular phylogenetic analyses (
This study is based on examination of herbarium specimens from the herbaria listed in the text, and comparison with a large (>5000) number of specimens of Solanum species from the African continent used in the preparation of our monograph on the prickly Solanum of continental Africa and Madagascar (
urn:lsid:ipni.org:names:820779-1
http://species-id.net/wiki/Solanum_rigidum
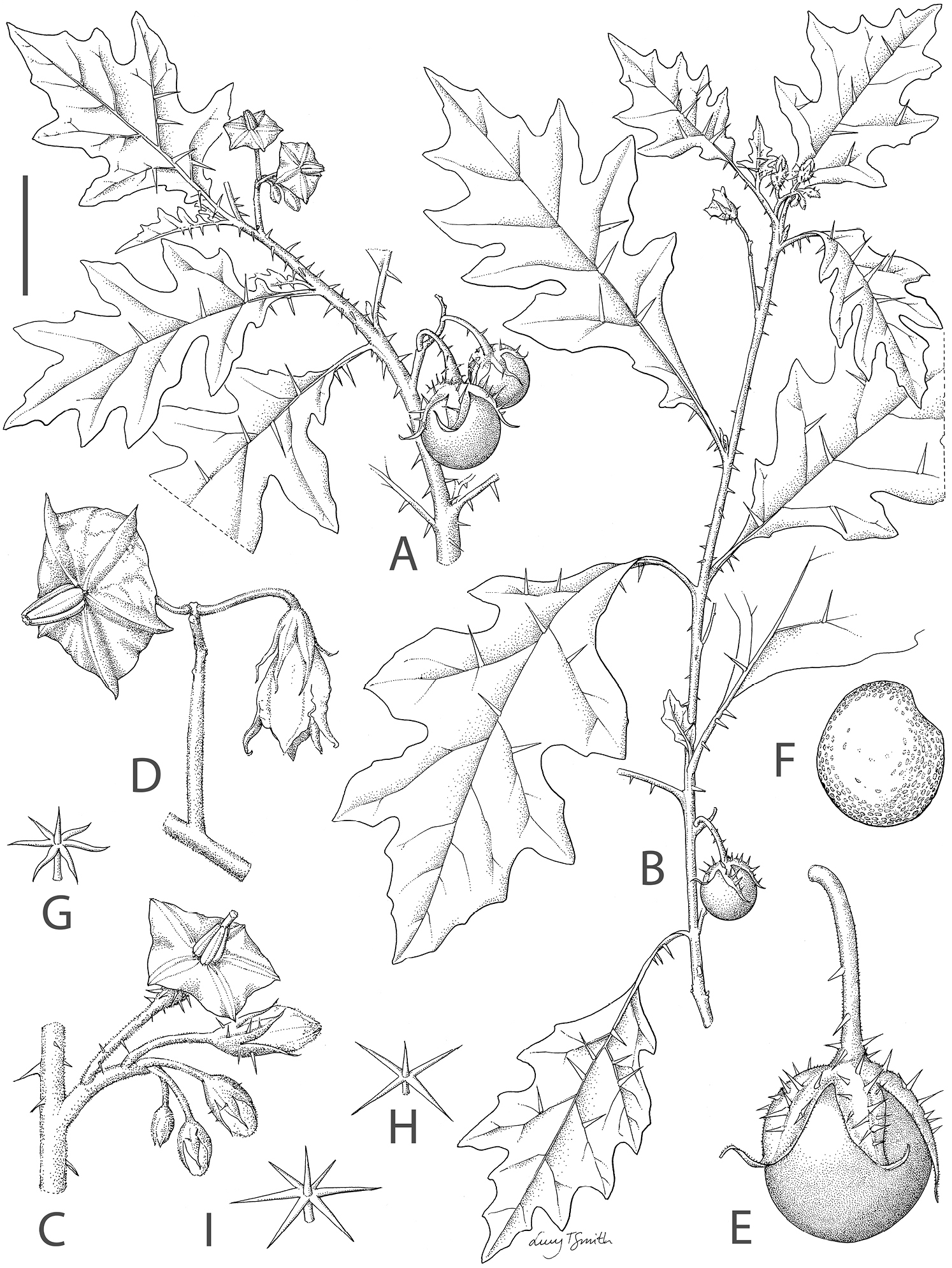
Figs 1–3Herbaceous subshrub to shrub, 0.6–1.5 m tall, armed; stems erect, densely (occasionally sparsely) pubescent with sessile or short-stalked translucent stellate trichomes < 0.5 mm long, the rays 4–6, ca. 0.5 mm long, the midpoints equal to the rays, prickly with straight to slightly curved broad-based prickles of varying lengths, 2–6 mm long, these sparsely stellate-pubescent; new growth densely stellate-pubescent, yellowish-brown in dry plants; bark of older stems dark greyish brown, not markedly glabrescent. Sympodial units difoliate and usually geminate, the leaves of a pair equal in size and shape. Leaves simple, 4.5–14(18) × 3.5–8.5(14) cm, elliptic, membranous to somewhat chartaceous, concolorous; upper surfaces sparsely pubescent with sessile (with some short-stalked) translucent stellate trichomes, the rays 3–4, ca. 0.3 mm long, the midpoint equal to the rays or occasionally somewhat longer; lower surfaces more densely pubescent with short-stalked or occasionally sessile translucent stellate trichomes to 0.3 mm long, the rays 4–5(-6), to 0.6 mm long, the midpoint equal to the rays, the lamina still easily visible; primary veins 3–5 pairs, drying yellowish, with 2–6 pale tan prickles to 6 mm long on both surfaces; base attenuate, usually decurrent onto the petiole; margins shallowly lobed, the lobes 3–4 on each side of the midrib, lobed 1/4–1/3 of the way to the midrib, the apices acute; apex acute; petioles usually somewhat winged from the decurrent leaf bases, 1.5–4.5 cm long, densely stellate-pubescent like the stems, usually with a few pale tan prickles of varying lengths, to 6 mm long, these straight or very slightly curved. Inflorescence 1–2.5 cm long, internodal (lateral) or opposite the leaves, simple or only once branched, with 5–6(10) flowers, the lowermost flower(s) hermaphroditic and the plants andromonoecious, densely pubescent with sessile or short-stalked translucent stellate trichomes with rays to 0.3 mm long like those of the stems; peduncle (0-)1–2 cm long, the lowermost flower often borne at the very base of the inflorescence; pedicels 1–1.5 cm long, 1–2 mm in diameter, densely stellate-pubescent like the rest of the inflorescence, that of the lowermost hermaphroditic flower usually somewhat stouter and usually with >10 pale tan prickles to 4 mm long, usually nodding at anthesis, articulated at the base; pedicel scars unevenly spaced 1–5 mm apart, the distance greatest between the lowermost flower and the rest. Buds elongate ellipsoid, the corolla approximately halfway exserted from the calyx before anthesis. Flowers 5-merous, strongly heterostylous, the lowermost (or lowermost 2–3) flower long-styled and hermaphroditic, usually slightly larger than the rest, the distal flowers short-styled and functionally male. Calyx tube 4–4.5 mm long, cup-shaped, the lobes 6–7 mm long, narrowly triangular with attenuate tips, densely pubescent with sessile and short-stalked stellate trichomes like those of the rest of the inflorescence, those of the hermaphroditic (long-styled) flowers usually with 5–25 yellowish prickles to 4 mm long. Corolla 2.5–3 cm in diameter, violet or white (type only), pentagonal with abundant interpetalar tissue, lobed less than 1/4 of the way to the base, the lobes 2–5 × 2–5 mm, usually with small acumens, planar at anthesis, glabrous adaxially, occasionally with a few stellate trichomes on the tips and margins, densely stellate-pubescent abaxially in a band 1.5 mm wide on either side of the petal midvein (exposed area in bud), the interpetalar tissue glabrous and thinner. Stamens equal, the filament tube ca. 0.5 mm long, the free portion of the filaments ca. 1.5 mm long, glabrous; anthers 6–7 × ca. 1.5 mm, usually slightly longer in hermaphroditic flowers, tapering, yellow, poricidal at the tips, the pores directed distally. Ovary densely pubescent with translucent stellate trichomes with 3–5 rays ca. 0.25 mm long; style 12–15 mm long in long-styled flowers, 1–1.5 mm long in short-styled flowers, sparsely pubescent with stellate trichomes like those of the ovary, these denser near the base; stigma expanded, bi-lobed. Fruit a globose berry, 2.5–3.5 cm in diameter, usually only 1 per infructescence, when immature mottled green and whitish green, maturing yellow, the pericarp leathery, ca. 1 mm thick, glabrous when mature; fruiting pedicels 2–2.5 cm long, 2–3 mm in diameter, thick and woody, pendent, pubescent and prickly as in the flowering pedicels; fruiting calyx splitting to the base, the lobes to 1 cm long, the tips usually reflexed at fruit maturity. Seeds > 100 per berry, 3–4 × 2.5–3 mm, reniform and slightly ovoid, not thickened at the margins, pale yellow or yellowish tan, the testal cells sinuate in outline. Chromosome number not known.
Habit of Solanum rigidum on the Cape Verde Islands. Photograph courtesy of MC Duarte.
Solanum rigidum. A habit with dense prickles and small deeply lobed leaves B habit with sparse prickles and larger more shallowly lobed leaves C inflorescence with a single lowermost hermaphroditic long-styled flower at the base D inflorescence with distal functionally male short-styled flowers E immature fruit and fruiting calyx with attenuate calyx lobe tips F seed G stellate trichome from the stem H, I stellate trichomes from the lower surface of the leaf. A, C, G–I drawn from Martins et al 468 B, F drawn from Barbosa & Silva 14072 D, E drawn from photographs of MC Duarte. Drawn by Lucy T. Smith.
Solanum rigidum on the Cape Verde Islands. A immature fruit with upturned sepal lobes, B functionally staminate flower with copious interpetalar tissue. Photographs courtesy of MC Duarte.
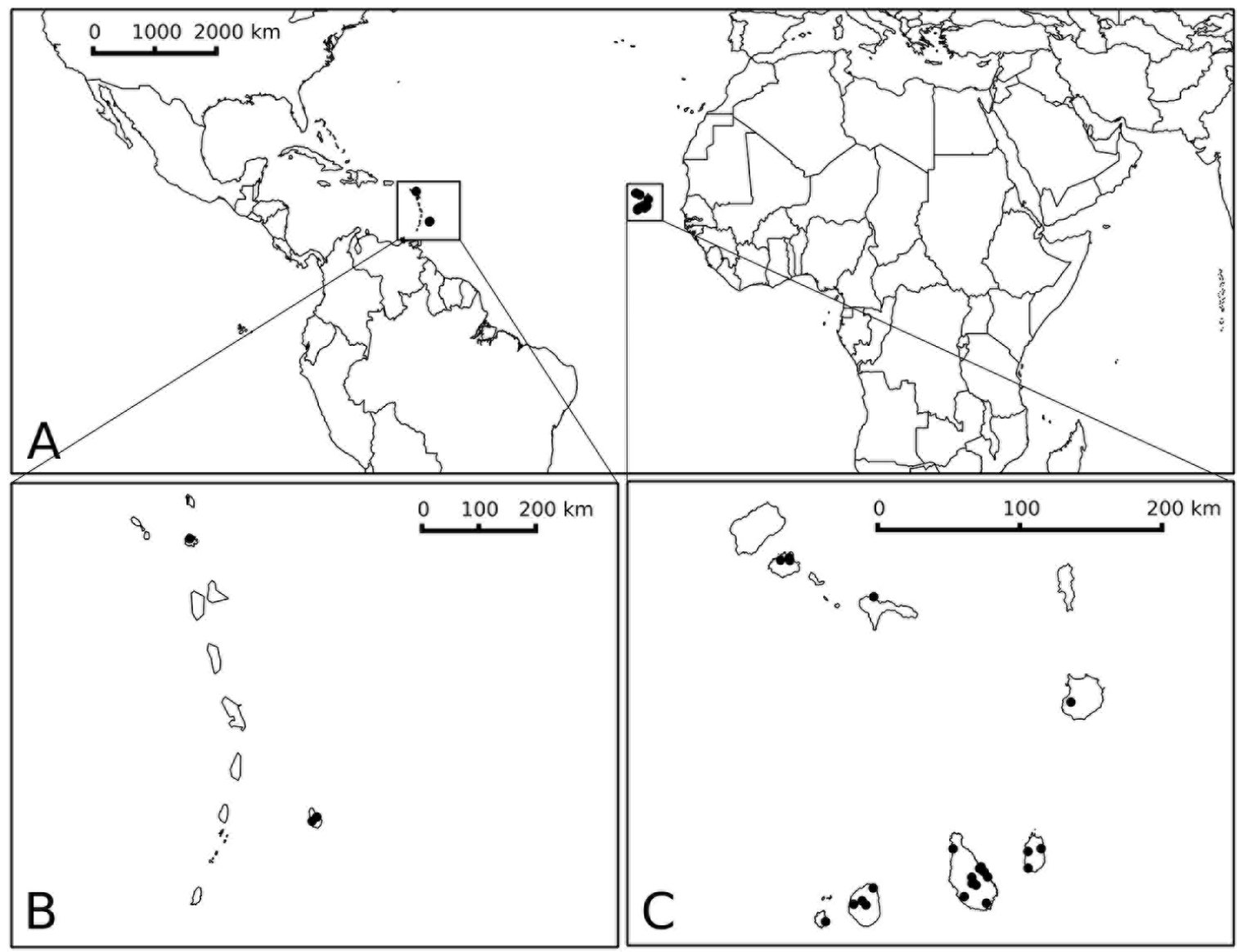
(Fig. 4) Endemic to the Cape Verde Islands, known from seven of the ten islands of the archipelago, on both the windward and leeward arcs; a few old collections from the Caribbean (see Discussion). Like most prickly solanum species, Solanum rigidum is a plant of disturbed and open areas and is somewhat weedy; it grows from sea level to 100 m.
Distribution map of Solanum rigidum. A global distribution in the Caribbean and the Cape Verdes B distribution on Antigua and Barbados C distribution on the Cape Verde Islands. Prepared by Paweł Ficinski.
Olho de vaca; olho de boi (cow’s eye, bull’s eye – perhaps in reference to the globose fruits).
Solanum rigidum has long been treated as Solanum fuscatum L. (
We have lectotypifed Solanum rigidum with a sheet in the Lamarck herbarium (P00357615, online at http://tinyurl.com/rigidum-LT) that is annotated with a reference to “illustr.”, the original place of publication (
In describing Solanum heteracanthum Dunal (1814) cited a specimen in the Richard herbarium (“Dunal, in herb. Rich.”) now held at P (P00344411). This is likely to be the holotype specimen but we prefer to designate this the lectotype as no specific sheet nor collector were cited in the protologue. This sheet is of a particularly prickly and possibly juvenile plant of Solanum rigidum; it has the characteristic elongate calyx lobes of this species.
A few collections from the Caribbean have confused understanding of the origins and distribution of this species; these plants may represent early introductions via the transatlantic routes used to transport the enslaved from Africa to the New World; these routes all passed through the Cape Verde archipelago from about 1560.
Morphologically, Solanum rigidum does not resemble any American species or species group of solanums, but it is more similar to African species of the Eggplant clade such as Solanum cerasiferum Dunal and Solanum campylacanthum A.Rich. in its greyish green leaves (Fig. 1), violet flowers and spherical fruits with tough pericarp that is green-striped when immature (see Fig. 3A) and yellow when ripe. Solanum rigidum differs from those species in its densely pubescent ovaries and long acuminate calyx lobes that are upturned in fruit (Fig. 3A). It shares with those taxa a strongly andromonoecious breeding system, with a single or few hermaphroditic flowers at the base of the inflorescence and the distal flowers with short styles and functioning as males (Fig. 3B). Andromonoecy is common in the spiny solanums (subgenus Leptostemonum Bitter) and is found in both New and Old World species.
Two other Solanum species occur on the Atlantic islands off the African coast (Macronesia): Solanum verspertilio Aiton and Solanum lidii Sunding, both from the Canary Islands. Both those species have strongly zygomorphic flowers with strongly unequal anthers while the flowers of Solanum rigidum are actinomorphic with anthers of equal size.
Preliminary DNA sequence data (S. Stern and M.S. Vorontsova pers. comm.) also indicate that Solanum rigidum is a member of the Eggplant clade, a large group of mostly East African taxa that includes the cultivated eggplant Solanum melongena L. Solanum rigidum may be of hybrid origin; in preliminary plastid analyses it is sister to Solanum campylacanthum while in trees based on the nuclear ITS region it is sister to Solanum macrocarpon L. (the gboma eggplant, a continental African species). The chromosome number of Solanum rigidum is not known, but Solanum campylacanthum is tetraploid in some parts of its range (see
The Cape Verde islands are geologically linked with the Canary Island archipelago (
The discovery that Solanum rigidum is not an introduction from the Americas but instead an endemic species in the Cape Verde islands highlights the need for conservation assessment on the islands in order to determine its range and population sizes. Solanum rigidum occurs on both of the main island groups of the Cape Verdes, on São Vincente, São Nicolau and Boa Vista of the Ilheus de Barlovento, and on Maio, Santiago, Fogo and Brava of the southern Ilheus de Sotovento (Fig. 4). Label data indicate Solanum rigidum occurs in disturbed habitats, often at the edges of washes and riverbeds, so it may be a weedy species despite its narrow geographic range and endemic status. Applying the IUCN criteria (
Cape Verde Islands. Boa Vista: sin. loc., 7 July 1934, A. Chevalier 44897 (P). Brava: Cachaço, Cova do Mar, 200 m, 29 October 1983, G. Cardoso de Matos 5434 (LISC); on the Ponton Road to the Fort, 26 March 1864, R.T. Lowe s.n. (P); Fogo: entre as povoações de Lomba e Ribeira Filipe, 900 m, 1 November 1983, G. Cardoso de Matos 5505 (LISC); San Filipe, 18 July 1934, A. Chevalier 44800 (P); Mosteiros, junto a pista de aviação, 10 m, 13 October 1991, Martins 468 (LISC); Curral da Chão, entre Achada Furna e Miguel Gonçalves, 15 October 1991, Martins 510 (LISC). Maio: Pedro Vaz, 17 May 1956, L.A. Grandvaux Barbosa 7437 (LISC); Calheta, 9 November 1964, J. Malato-Beliz 141 (LISC); Pedro Vaz, 11 November 1964, J. Malato-Beliz 244 (LISC); Vila da Maia, Dunas de Morrinho, 17 November 1964, J. Malato-Beliz 360 (LISC). Santiago: São Jorge dos Orgãos, Ribeirão Galinha, 350 m, 22 October 1983, G. Cardoso de Matos 5304 (LISC); Ribeira Grande de Santiago, a longo de leito seco da Ribeira de Fundão depois de passar os regadios que estão junto as casas, 540 m, 15 July 1993, M.C. Duarte 540 (LISC); estrada Praia-Tarrafal, 1.1 km depois Porto Fundo, 65 m, 9 December 1955, L.A. Grandvaux Barbosa 5909 (LISC); Santa Cruz, aluviões do Ribeira da Cruz, 16 November 1982, L.A. Grandvaux Barbosa 14072 (LISC); Ribeira de Santa Cruz, 20 March 1983, L.A. Grandvaux Barbosa 14486 (LISC); between S. Domingo and Os Orgãos, 31 January 1866, R.T. Lowe s.n. (BM, LE); Villa do Praia, January 1861, F.M.J. Welwitsch 6086 (BM); Pedra Badejo, 10 m, 17 October 1992, M.C. Duarte 56 (LISC); Chã de Vaca, 277 m, 16 October 1994, M.C. Duarte 701 (LISC); Foz da Ribeira de Mangue, 30 m, 11 July 1993, M.C. Duarte 502 (LISC); Baia de Chão Bom, 2 m, 23 October 1994, M.C. Duarte 1189a (LISC); sin. loc, C. Peters s.n. (LE). São Nicolau: am Weg von Estancia Bras zum Ribeira Quameros, 50 m, 3 January 1986, N. Kilian 1014 (B). São Vicente: Monte Verde, September 1934, A. Chevalier 45744 (P); Porto Grande, 22 November 1894, E.H.L. Krause 17687 (B); ascent of Monte Verde, 609 m, 5 January 1866, R.T. Lowe s.n. (BM).
Antigua and Barbuda. Antigua: sin. loc, Anonymous 16 (K).
Barbados. Barbados: Foster Hall Spring, January 1890, H.F.A. Eggers 7226 (P); near Welches, St. Thomas, June 1935, A.C.S. McIntosh 195 (P); Bathsheba and Hastings, April 1895, J.F. Waby 53a (BM, K).
This work was funded by the National Science Foundation Planetary Biodiversity Inventory (PBI) Solanum: A worldwide treatment (DEB-0316614). We thank Maria Cristina Duarte (LISC) for photographs and facilitating the loan of Cape Verde material. We are also grateful for Lucy Smith for the artwork and to Paweł Ficinski for cartography.