(C) 2013 Amanda Lee Grusz. This is an open access article distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
Citation: Grusz AL, Windham MD (2013) Toward a monophyletic Cheilanthes: The resurrection and recircumscription of Myriopteris (Pteridaceae). PhytoKeys 32: 49–64. doi: 10.3897/phytokeys.32.6733
The fern genus Cheilanthes (Pteridaceae) has perplexed taxonomists for more than two centuries. Complex patterns of evolution involving rampant morphological convergence, polyploidy, hybridization, and apomixis have made the taxonomy of this group especially difficult. Fortunately, recent phylogenetic analyses have helped to clarify relationships among cheilanthoid taxa. Based on these findings, we here formalize an updated taxonomy for one monophyletic clade comprising 47 primarily North and Central American taxa usually included in Cheilanthes. Because the type species of Cheilanthes (Cheilanthes micropteris) is only distantly related to this clade, we resurrect the genus Myriopteris to accommodate these taxa, and present a revised circumscription for the group, including 36 new combinations.
Cheilanthes, cheilanthoid, myriopterid, myriopteroid, nomenclature, taxonomy
A “practical and natural” generic classification of cheilanthoid ferns (Pteridaceae) has eluded taxonomists for more than 200 years and was viewed by
Here, we focus on the primarily New World lineage previously referred to as the “American Cheilanthes” (
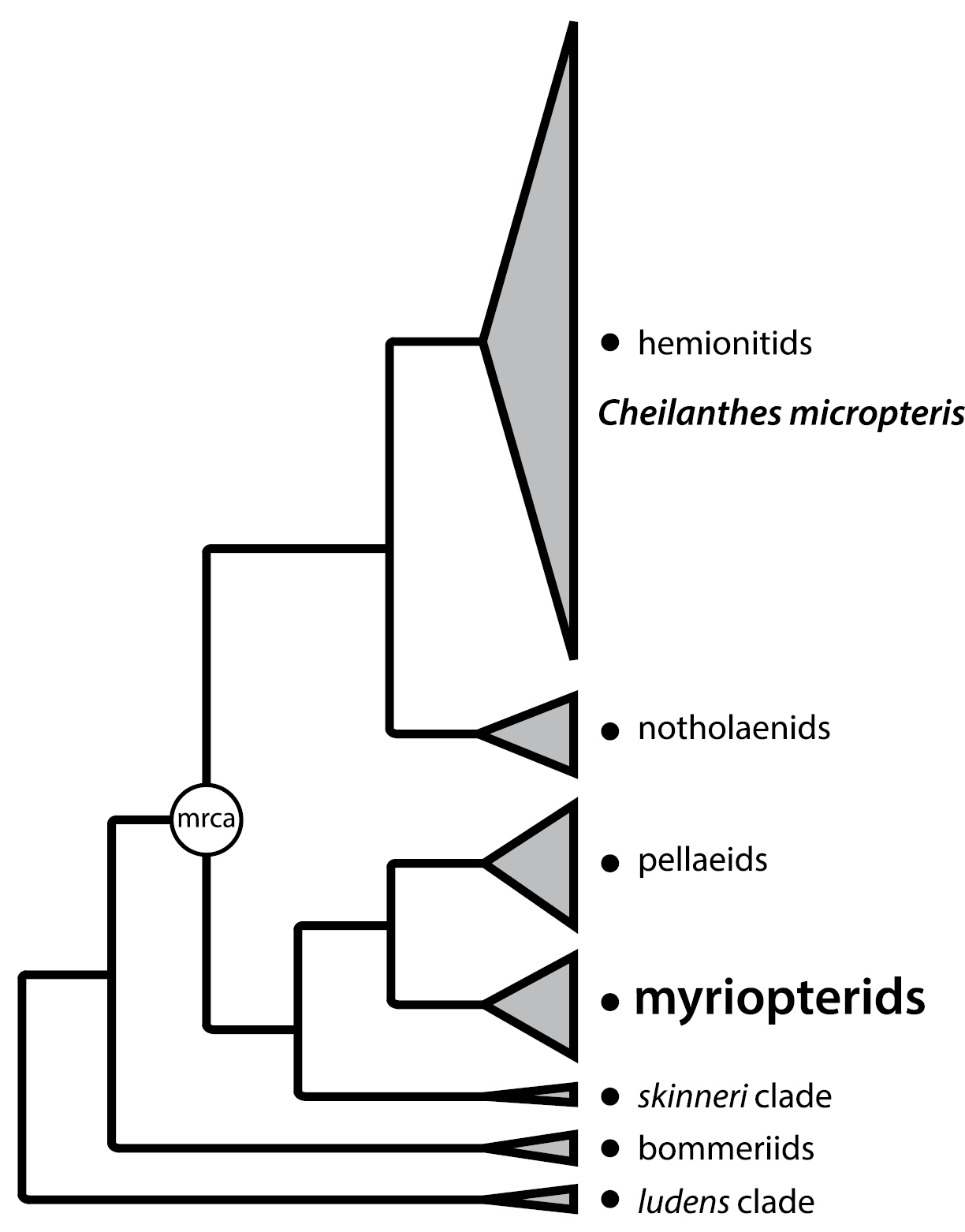
Summary phylogeny for cheilanthoid ferns, indicating the placement of Cheilanthes micropteris (the type species for Cheilanthes) within the hemionitid clade—only distantly related to the myriopterid clade. The six major clades of cheilanthoid ferns are shown with tips roughly proportional to clade size. The most recent common ancestor (mrca) of Cheilanthes micropteris and the myriopterid clade is indicated. Modified with permission from
When any species or clade is removed from Cheilanthes, the first issue that must be addressed involves their relationship to Allosorus pusillus (Willd. ex Bernh.) Bernh. [= Cheilanthes pteridioides (Reich.) C. Chr.]. This species was designated the lectotype of Allosorus Bernh. by
One potentially viable option for generic placement of the myriopterid clade would be to include it within a revised circumscription of Pellaea Link. All recent phylogenetic studies with adequate sampling of the two groups (e.g.,
If the expansion of Pellaea is ruled out, there remain three other generic names typified by species belonging to the myriopterid clade: 1) Myriopteris, described by
The original concept of Myriopteris (
Although this “microphyllous” leaf morphology is common within Myriopteris, it does not characterize the entire clade (
Ideally, morphological and/or cytological synapomorphies would substantiate phylogenetic relationships inferred from DNA sequence data. However, easily observed synapomorphies distinguishing the various clades of cheilanthoid ferns are few, and homoplastic characters abound. To paraphrase
Molecular analyses spanning the diversity of cheilanthoid species (Windham et al. unpublished) illuminate one particularly useful character distinguishing Myriopteris, as defined herein, from Cheilanthes s.s. The taxa most closely related to the type species of the latter [Cheilanthes micropteris plus all Australian Cheilanthes and a group of South American species including the Cheilanthes scariosa (Sw.) C. Presl complex of
Myriopteris marsupianthes Fée, Mém. Fam. Foug. 5: 149, t. 12A. f. 1. 1852
Plants rupestral or terrestrial. Rhizomes compact to long-creeping, ascending or horizontal, scaly. Rhizome scales lanceolate to acicular, concolorous (tan to dark brown) or bicolorous (with dark central stripe and brown margins). Leaf vernation non-circinate to circinate. Petiolescastaneous to black, scaly and/or pubescent, rarely almost glabrous. Rachises terete or flattened or grooved adaxially, with indument similar to that of the petioles. Blades2- to 4-pinnate (rarely pinnate-pinnatifid), lanceolate to ovate-deltate, occasionally linear or pentagonal; adaxial surfaces glabrous or pubescent; abaxial surfaces scaly and/or pubescent or rarely glabrous. Ultimate segments round to oblong-ovate, minute to >1 cm long, the veins obscure and not ending in prominent hydathodes. Segment margins usually recurved, with a poorly differentiated false indusium (strongly differentiated in Myriopteris lendigera and Myriopteris marsupianthes). Sori usually partly to completely covered by the recurved segment margins, the sporangia clustered at vein tips. Sporangia 64-spored (in sexual species) or 32-spored (in apomicts). Spores globose-tetrahedral, tan to brown, cristate to rugulate. Chromosome numbers n = 29, 30, 58, 60 (sexual species); n = 2n = 87, 90 (apomictic triploids); n = 2n = 120 (apomictic tetraploids).
Species of Myriopteris range from southern Canada through the Caribbean and Central America to southern Chile, with one species (Myriopteris rawsonii) endemic to Namibia and South Africa. Mexico is the center of species diversity for the genus; 34 of the 44 species can be found in Mexico, and seven of these are endemic.
1) Myriopteris aemula (Maxon) Grusz & Windham, comb. nov. Cheilanthes aemula Maxon, Contr. U.S. Natl. Herb. 10: 495. 1908. Type: Mexico. Tamaulipas: Victoria, in river canyon, under overhanging rocks, altitude about 320 meters, February 1 to April 9, 1907, Palmer 187 (holotype: US; isotype: US). urn:lsid:ipni.org:names:77134841-1
2) Myriopteris alabamensis(Buckley) Grusz & Windham, comb. nov. Pteris alabamensis Buckley, Amer. J. Sci. Arts 45: 177. 1843. Cheilanthes alabamensis (Buckley) Kunze, Linnaea 20: 4. 1847. Type: USA. Alabama: Growing in tufts on limestone rocks that form the banks of the Tennessee River, at the foot of Muscle Shoals, Buckley s.n. (holotype: PH; isotypes: MO, NY). urn:lsid:ipni.org:names:77134842-1
3) Myriopteris allosuroides (Mett.) Grusz & Windham, comb. nov. Cheilanthes allosuroides Mett., Abh. Senckenberg. Naturf. Ges. 3: 78. 1859. Pellaea allosuroides (Mett.) Hieron., Hedwigia 62: 18. 1920. Type: Mexico, Schmitz s.n. (holotype: location unknown). urn:lsid:ipni.org:names:77134843-1
4) Myriopteris aurea (Poir.) Grusz & Windham, comb. nov. Pteris aurea Poir. Encyclopédie Méthodique, Botanique 5: 710. 1804. Type: Peru. Elle a été recueillie au Pérou par Joseph de Jussieu s.n. (sheet 1333 in hb. Jussieu; holotype: P). urn:lsid:ipni.org:names:77134844-1
Acrostichum bonariense Willd., Sp. Pl., ed. 4, 5(1): 114. 1810. Notholaena bonariensis (Willd.) C. Chr., Index Filic. 459. 1906. Cheilanthes bonariensis (Willd.) Proctor, Bull. Inst. Jamaica, Sci. Ser. 5: 15. 1953.
In Cheilanthes, this has been called Cheilanthes bonariensis (Willd.) Proctor because use of the oldest applicable epithet (based on Pteris aurea Poir.) was blocked by the earlier publication of Cheilanthes aurea Baker (
5) Myriopteris chipinquensis (Knobloch & Lellinger) Grusz & Windham, comb. nov. Cheilanthes chipinquensis Knobloch & Lellinger, Amer. Fern J. 59: 8. 1969. Type: Mexico. Nuevo Leon: Chipinque Mesa, outside Monterey, Knobloch 1996B (holotype: MSC; isotypes: F, GH, MEXU, MICH, UC, US). urn:lsid:ipni.org:names:77134845-1
6) Myriopteris cinnamomea(Baker) Grusz & Windham, comb. nov. Notholaena cinnamomea Baker in Hook. & Baker, Syn. Fil. ed. 2. 515. 1874. Cheilanthes cinnamomea (Baker) Domin., Biblioth. Bot. 20: 133. 1913. hom. illeg. non Cheilanthes cinnamomea D. C. Eaton, Proc. Amer. Acad. Arts 18: 186. 1883. Type: Guatemala. Mo[n]tagua, 1862, Salvin & Goodman s.n. (holotype: K; isotype: BM). urn:lsid:ipni.org:names:77134870-1
Cheilanthes tryonii T. Reeves, Brittonia 32: 504. 1980.
In Cheilanthes, this species has been called Cheilanthes tryonii T. Reeves because use of the oldest applicable epithet (based on Notholaena cinnamomea Baker) was blocked by the earlier publication of Cheilanthes cinnamomea D. C. Eaton (
7) Myriopteris clevelandii (D. C. Eaton) Grusz & Windham, comb. nov. Cheilanthes clevelandii D. C. Eaton, Bull. Torrey Bot. Club 6: 33. 1875. Type: USA. California: Growing on a mountain about forty miles from San Diego at an elevation of about 2500 feet, Cleveland s.n. (holotype: YU; isotypes: GH, P, US). urn:lsid:ipni.org:names:77134846-1
8) Myriopteris cooperae (D. C. Eaton) Grusz & Windham, comb. nov. Cheilanthes cooperae D. C. Eaton, Bull. Torrey Bot. Club 6: 33. 1875. Type: USA. California: near Santa Barbara, Mrs. Ellwood Cooper (syntype: YU); Sierra Valley, Lemmon s.n. (syntype: YU). urn:lsid:ipni.org:names:77134847-1
9) Myriopteris covillei (Maxon) Á. Löve & D. Löve, Taxon 26: 325. 1977. Cheilanthes covillei Maxon, Proc. Biol. Soc. Wash. 31: 147. 1918. Type: USA. California: Surprise Canyon, Panamint Mountains, 13 April 1891, 1550 meters, Coville & Funston 593 (holotype: US). urn:lsid:ipni.org:names:77134848-1
10) Myriopteris cucullans (Fée) Grusz & Windham, comb. nov. Cheilanthes cucullans Fée, Mém. Fam. Foug. 7: 39, t. 25, f. 4. 1857. Type: Mexico, ad vallem Mexicanum, Schaffner 82 [holotype: RB; isotypes: K, US (fragment)]. urn:lsid:ipni.org:names:77134873-1
11) Myriopteris fendleri (Hook.) E. Fourn., Mex. Pl. 1: 125. 1872. Cheilanthes fendleri Hook., Sp. Fil. 2: 103, p. 107b. 1852. Type: USA. New Mexico, 1847, Fendler 1015 [holotype: K; isotypes: GH, MO, NY, US (fragment)].
12) Myriopteris × fibrillosa(Davenp.) Grusz & Windham, comb. nov. Cheilanthes lanuginosa var. fibrillosa Davenp., Bull. Torrey Bot. Club 12: 21. 1885. Cheilanthes fibrillosa (Davenp.) Davenp., Bull. Torrey Bot. Club 15: 225. 1888. Type: USA. California: San Jacinto Mountains, June 1882, Parish & Parish s.n. (holotype: GH). urn:lsid:ipni.org:names:77134880-1
13) Myriopteris fimbriata (A. R. Sm.) Grusz & Windham, comb. nov. Cheilanthes microphylla (Sw.) Sw. var. fimbriata A. R. Sm., Amer. Fern J. 70: 19, 21., f. 9–10. 1980. Type: Mexico. Chiapas: Munic. Frontera Comalapa, 6–8 km east of Frontera Comalapa, Breedlove 39018 (holotype: DS). urn:lsid:ipni.org:names:77134881-1
Cheilanthes fimbriata (A. R. Sm.) Mickel & Beitel, Mem. New York Bot. Gard. 46: 112. 1988. hom. illeg., non Cheilanthes fimbriata Vis., Fl. Dalmat. 1. 42 t. 1 f. 1. 1842.
14) Myriopteris gracilis Fée, Mém. Fam. Foug. 5: 150, t. 29, f. 6. 1852. Cheilanthes gracilis (Fée) Mett. ex Riehl, Abh. Senckenberg. Naturf. Ges. 80. 1859. hom. illeg., non Cheilanthes gracilis (Michx.) Kaulf., Enum. Filic. 209. 1824. Type: USA. Missouri: Jefferson County, Habitat ad rupes circa Hillsboro, Americâ septentr., Riehl 529 (isotypes: MO, US).
Cheilanthes feei T. Moore, Index Fil., 38. 1857.
Myriopteris lanuginosa J. Sm. Hist. Fil. 280. 1875. [non M. lanuginosa (Mart. & Gal.) E. Fourn. Mexic. Pl. 1: 125. 1872.]
In Cheilanthes, this has been called Cheilanthes feei T. Moore because use of the oldest applicable epithet (based on Myriopteris gracilis Fée) was blocked by the earlier publication of Cheilanthes gracilis (Michx.) Kaulf. With the transfer of this species to Myriopteris, we revert to the original name published by Fée in 1852.
15) Myriopteris gracillima (D. C. Eaton) J. Sm., Hist. Fil. 280. 1875. Cheilanthes gracillima D. C. Eaton, Rep. U.S. Mex. Bound. Botany 2: 234. 1859. Type: USA. Oregon: Cascade Mountains, 7000 feet of altitude, latitude 44°, Bigelow s.n. (lectotype: YU).
16) Myriopteris intertexta (Maxon) Grusz & Windham, comb. nov. Cheilanthes covillei Maxon subsp. intertexta Maxon, Proc. Biol. Soc. Wash. 31: 149. 1918. Cheilanthes intertexta (Maxon) Maxon in Abrams, Ill. Fl. Pacific States 1: 28. 1923. Type: USA. California: Santa Clara County, Santa Cruz Mountains, collected at the top of Black Mountain, 6 July 1903, Dudley s.n. (holotype: DS). urn:lsid:ipni.org:names:77134849-1
17) Myriopteris jamaicensis (Maxon) Grusz & Windham, comb. nov. Cheilanthes jamaicensis Maxon, Contr. U.S. Natl. Herb. 24: 51. 1922. Type: Jamaica. Below Cinchona, 28 February 1919, Harris 12905 (holotype: US; isotypes: GH, MO, NY). urn:lsid:ipni.org:names:77134850-1
18) Myriopteris lanosa (Michx.) Grusz & Windham, comb. nov. Nephrodium lanosum Michx. Fl. Bor.-Amer. 2: 270. 1803. Cheilanthes lanosa (Michx.) D. C. Eaton, Rep. U.S. Mex. Bound., Botany 2: 234. 1859. Type: USA. Tennassee (sic) et Carolinae septentrionalis (non designatus). urn:lsid:ipni.org:names:77134851-1
Myriopteris vestita (Sw.) J. Sm., Cul. Ferns 29. 1857. (fide C. Chr. 1906.) Adiantum vestitum Spreng., Anleit. Kenntn. Gew. 3: 122. 1804.
19) Myriopteris lendigera (Cav.) Fée, Mém. Fam. Foug. 5: 149. 1852 (as Myriopteris lentigera). Pteris lendigera Cav., Descr. Pl. 268. 1801. Cheilanthes lendigera (Cav.) Sw., Syn. Fil. 128, 328. 1806. Type: Mexico. Hidalgo: Ixmiquilpan en la Nueva España, Nee s.n. [syntype: MA, US (fragment)]; Ecuador. Bolivar: junto á Guaranda en el Reyno de Quito, Nee s.n. (syntype: MA).
Cheilanthes minor Mart. & Gal. Mém. Act. Brux. 75, pl. 21, f. 1. 1842. Myriopteris minor (Mart. & Gal.) Fée, Mém. Fam. Foug. 5: 150. 1852.
Cheilanthes lanuginosa Mart. & Gal. Mém. Act. Brux. 75, pl. 20, f. 2. 1842. Myriopteris lanuginosa (Mart. & Gal.) E. Fourn. Mex. Pl. 1: 125. 1872.
Myriopteris villosa Fée, Mém. Fam. Foug. 5: 149. t. 28, f. 1. 1852.
Cheilanthes frigida Linden ex T. Moore, Gard. Chr. 772. 1857. Myriopteris frigida (Linden ex T. Moore) J. Sm. Cat. Cult. Ferns 28. 1857.
Myriopteris lendigera (Cav.) J. Sm., Cat. Cult. Ferns 28. 1857. hom. illeg.
Pomataphytum pocillatum M. E. Jones, Contributions to Western Botany 16: 12. 1930.
20) Myriopteris lindheimeri (Hook.) J. Sm., Bot. Voy. Herald. 340. 1856. Cheilanthes lindheimeri Hook., Sp. Fil. 2: 101, t. 107a. 1852. Type: USA. Western Texas, 1847, Lindheimer 744 [lectotype: K; isolectotypes: GH, P (2 sheets), SD, US, YU].
21) Myriopteris longipila (Baker) Grusz & Windham, comb. nov. Cheilanthes longipila Baker, Ann. Bot. (Oxford) 5: 211. 1891. Type: Mexico. San Luis Potosí, 22°N Lat., 6000–8000 ft., Parry & Palmer 989 [holotype: K; isotype: US (fragment)]. urn:lsid:ipni.org:names:77134852-1
22) Myriopteris longipila subsp. brevipila (Mickel) Grusz & Windham, comb. nov. Cheilanthes longipila var. brevipila Mickel, Mem. New York Bot. Gard. 88: 198–199, f. 84N–Q, 87J–M. 2004. Type: Mexico. Guerrero: 2 km al SE de Amatitlán, 1600 m, 13 August 1994, Soto 1052 (holotype: NY; isotype: FCME). urn:lsid:ipni.org:names:77134882-1
23) Myriopteris marsupianthes Fée, Mém. Fam. Foug. 5: 149, t. 12A, f. 1. 1852. Cheilanthes marsupianthes (Fée) T. Reeves ex Mickel & A. R. Sm. Mem. New York Bot. Gard. 88: 201, f. 83M–P. 2004.Type: Mexico. Veracruz: Pic d’Orizaba, Martens & Galeotti 6256 (holotype: P; isotype: BR).
24) Myriopteris maxoniana (Mickel) Grusz & Windham, comb. nov. Cheilanthes maxoniana Mickel, Mem. New York Bot. Gard. 88: 201, f. 87A–D. 2004. Type: Mexico. Tamaulipas: San Lucas, Viereck 76 (holotype: US). urn:lsid:ipni.org:names:77134853-1
25) Myriopteris mexicana (Davenp.) Grusz & Windham, comb. nov. Cheilanthes mexicana Davenp., Bull. Torrey Bot. Club 15: 227. 1888. Type: Mexico. Chihuahua: on the verge of a high cliff near the summit of Potrero Peak (Santa Eulalia Mts.), October 1886, 7300 ft., Pringle 827 (holotype: GH; isotypes: MO, BR, DS, NY, P, UC, US, YU). urn:lsid:ipni.org:names:77134854-1
26) Myriopteris mickelii (T. Reeves) Grusz & Windham, comb. nov. Cheilanthes mickelii T. Reeves, Brittonia 32: 502, f. 1–5. 1980. Type: Mexico. Oaxaca: Distr. Yautepec, Mickel 4210 (holotype: NY; isotypes: MO, UC). urn:lsid:ipni.org:names:77134855-1
27) Myriopteris microphylla (Sw.) Grusz & Windham, comb. nov. Adiantum microphyllum Sw., Prodr. 135. 1788. Cheilanthes microphylla (Sw.) Sw., Syn. Fil. 127. 1806. Type: Jamaica, Swartz s.n. (holotype: S). urn:lsid:ipni.org:names:77134856-1
28) Myriopteris moritziana (Kunze) Grusz & Windham, comb. nov. Cheilanthes moritziana Kunze, Linnaea 23: 307. 1850. Type: Venezuela. Caracas: La Guayra, Moritz 263 (lectotype: B; isolectotype: GH). urn:lsid:ipni.org:names:77134857-1
29) Myriopteris myriophylla (Desv.) J. Sm., Bot. Voy. Herald, 340. 1856. Cheilanthes myriophylla Desv., Ges. Naturf. Freunde Berlin Mag. Neuesten Entdeck. Gesammten Naturk. 5: 328. 1811. Type: South America. Anon. s.n. (holotype: P).
Cheilanthes elegans Desv. Ges. Naturf. Freunde Berlin Mag. 5: 328. 1811. Myriopteris elegans (Desv.) J. Sm., Cat. Cult. Ferns 29. 1857.
Cheilanthes paleacea Mart. & Gal., Mém. Foug. Mexique 76, pl. 21, f. 2. 1842. Myriopteris paleacea (Mart. & Gal.) Fée, Mém. Fam. Foug. 5: 149, t. 29, f. 6. 1852.
Myriopteris intermedia E. Fourn., Bull. Soc. Bot. Fr. 27: 328. 1880. hom. illeg., non Fée, Mém. Fam. Foug. 5: 149. 1852.
30) Myriopteris newberryi(D. C. Eaton) Grusz & Windham, comb. nov. Notholaena newberryi D. C. Eaton, Bull. Torrey Bot. Club 4: 12. 1873. Cheilanthes newberryi (D. C. Eaton) Domin, Biblioth. Bot. 20: 133. 1913. Types: USA. California: San Diego, 9 November 1857, Newberry 1352 (syntype: MO, YU); San Diego, 1866, Wood s.n. (syntype: YU); Southern California: S. W. corner of San Bernardino County, rocks in the Temescal range, 22 January 1861, W. H. Brewer s.n. (syntype: YU). urn:lsid:ipni.org:names:77134858-1
31) Myriopteris notholaenoides (Desv.) Grusz & Windham, comb. nov. Pteris notholaenoides Desv., Mém. Soc. Linn. Paris 6: 299. 1827. Cheilanthes notholaenoides (Desv.) Maxon ex Weath., Contr. Gray Herb. 114: 34. 1936. Type: Hispaniola, Anon. s.n. (holotype: P). urn:lsid:ipni.org:names:77134859-1
32) Myriopteris × parishii (Davenp.) Grusz & Windham, comb. nov. Cheilanthes parishii Davenp., Bull. Torrey Bot. Club 8: 59. 1881. Type: USA. California: San Diego County, W. J. Parish s.n. (holotype: GH; isotypes: GH, YU). urn:lsid:ipni.org:names:77134860-1
33) Myriopteris parryi (D. C. Eaton) Grusz & Windham, comb. nov. Notholaena parryi D. C. Eaton, Amer. Naturalist 9: 351. 1875. Cheilanthes parryi (D. C. Eaton) Domin, Biblioth. 85: 133. 1913. Type: USA. Utah: C. C. Parry 263 (holotype: YU; isotypes: GH, US, YU). urn:lsid:ipni.org:names:77134861-1
34) Myriopteris peninsularis (Maxon) Grusz & Windham, comb. nov. Cheilanthes peninsularis Maxon, Contr. U.S. Natl. Herb. 10: 496. 1908. Type: Mexico. Baja California, T. S. Brandegee s.n. (holotype: US). urn:lsid:ipni.org:names:77134862-1
35) Myriopteris peninsularissubsp. insularis (Weath.) Grusz & Windham, comb. nov. Cheilanthes peninsularis (Maxon) var. insularis Weath., Amer. Fern J.21: 25. 1931. Type: Mexico. Socorro Island, Mason 1616 (holotype: CAS). urn:lsid:ipni.org:names:77134884-1
36) Myriopteris pringlei (Davenp.) Grusz & Windham, comb. nov. Cheilanthes pringlei Davenp., Bull. Torrey Bot. Club 10: 61, t. 34. 1883. Type: USA. Arizona: C. G. Pringle s.n. (holotype: GH; isotypes: DS, MO, NY, US, YU). urn:lsid:ipni.org:names:77134863-1
37) Myriopteris pringlei subsp. moncloviensis (Baker) Grusz & Windham, comb. nov. Cheilanthes moncloviensis Baker, Ann. Bot. (Oxford) 5: 210. 1891. Cheilanthes pringlei var. moncloviensis (Baker) Mickel, Mem. New York Bot. Gard. 88: 207–208, f. 79J–M. 2004. Type: Mexico. Coahuila: Soledad, E. Palmer 1378 (holotype: K; isotypes: MO, NY, US). urn:lsid:ipni.org:names:77134864-1
38) Myriopteris rawsonii(Mett. ex. Kuhn) Grusz & Windham, comb. nov. Cheilanthes rawsonii Mett. ex. Kuhn, Filices Africanae 75. 1868. Type: Africa. Cape Province: Namaqualand, between Specktakel and Komaggas, Whitehead s.n. (holotype: BM; isotype: K). urn:lsid:ipni.org:names:77134878-1
39) Myriopteris rufaFée, Mém. Fam. Foug. 8: 77. 1857. Type. Mexico. Veracruz: Volcan de Orizaba, Schaffner 83 (holotype: P?; isotype: RB?).
Cheilanthes eatonii Baker in Hook. & Baker, Syn. Fil. 140. 1867.
Cheilanthes castanea Maxon, Proc. Biol. Soc. Wash. 32: 111. 1919.
In Cheilanthes, this has been called Cheilanthes eatonii Baker. Examination of putative type specimens of Myriopteris rufa housed at RB (digital image) and P indicates that the latter name very likely represents the same species as broadly defined by recent authors (e.g.,
40) Myriopteris scabra (C. Chr.) Grusz & Windham, comb. nov. Pellaea scabra C. Chr., Index Filic. 483. 1906. Type: USA. Texas: crevices of rock on hills, Turkey Creek, 25 June 1849, Wright 824 (holotype: K; isotypes: GH, NY, US).
Cheilanthes aspera Hook., Sp. Fil. 2: 111, t. 108A. 1852. hom. illeg., non Cheilanthes aspera Kaulf., Linnaea 6(1): 186. 1831. urn:lsid:ipni.org:names:77134865-1
Cheilanthes horridula Maxon, Amer. Fern J. 8: 94. 1918.
In Cheilanthes, this has been called Cheilanthes horridula Maxon because use of the oldest legitimate epithet (based on Pellaea scabra C. Chr.) was blocked by the earlier publication of Cheilanthes scabra H. Karst. (Maxon 1918). With the transfer of this species to Myriopteris, we revert to the older, exceedingly appropriate epithet.
41) Myriopteris tomentosa (Link) Fée, Mém. Fam. Foug. 5: 149. 1852. Cheilanthes tomentosa Link, Hort. Berol. 2: 42. 1833. Type: Mexico. Anon. s.n. [holotype: B; isotypes: PH, US (fragment)].
Cheilanthes bradburii Hook., Sp. Fil. 2: 97, t. 109b. 1852. Myriopteris bradburii (Hook.) J. Sm. Hist. Fil. 280. 1875.
42) Myriopteris viscida(Davenp.) Grusz & Windham, comb. nov. Cheilanthes viscida Davenp., Bull. Torrey Bot. Club 6: 191. 1877. Types: USA. California: Eastern slope of the Sierra Nevada near San Gogorio Pass, April 1876, Parry & Lemmon 427 (syntype: NY); California/Nevada: Downieville Buttes and bluffs of White Water River on the Colorado Desert, April–May, Lemmon s.n. (syntype: NY). urn:lsid:ipni.org:names:77134866-1
43) Myriopteris windhamii Grusz, Amer. Fern J. 103: 113. 2013. Type: USA. Arizona: Huachuca Mountains, Windham 4165 (holotype: DUKE; isotypes: ARIZ, ASC, ASU, GH, MO, NMC, NY, TEX/LL, UNM, US, UT).
Cheilanthes villosa Davenp. ex Maxon, Proc. Biol. Soc. Wash. 31: 142. 1918.
In Cheilanthes, this has been called Cheilanthes villosa Davenp. ex Maxon. Because transfer of the epithet villosa to Myriopteris is blocked by the earlier publication of Myriopteris villosa Fée (= Myriopteris lendigera fide Reeves 1979), we use the replacement name for this distinctive taxon published by Grusz (2013).
44) Myriopteris wootonii (Maxon) Grusz & Windham, comb. nov. Cheilanthes wootonii Maxon, Proc. Biol. Soc. Wash. 3: 146. 1918. Type: USA. Arizona: Santa Rita Mountains, Wooton s.n. (holotype: US). urn:lsid:ipni.org:names:77134867-1
45) Myriopteris wrightii (Hook.) Grusz & Windham, comb. nov. Cheilanthes wrightii Hook., Sp. Fil. 2: 87, t. 110A. 1858. Type: USA. Texas–New Mexico: Wright 823 (holotype: K; isotypes: GH, NY, US). urn:lsid:ipni.org:names:77134868-1
46) Myriopteris yatskievychiana (Mickel) Grusz & Windham, comb. nov. Cheilanthes yatskievychiana Mickel, Mem. New York Bot. Gard. 88: 212–213, f. 74F–K. 2004. Type: Mexico. Sonora: Sierra del Aliso, A. Búrquez M. 96-302 (holotype: MO). urn:lsid:ipni.org:names:77134869-1
47) Myriopteris yavapensis (T. Reeves ex Windham) Grusz & Windham, comb. nov. Cheilanthes yavapensis T. Reeves ex Windham, Contr. Univ. Michigan Herb. 19: 32. 1993. Type: USA. Arizona: Yavapai County, Windham 202 (holotype: UT; isotypes: ASC, ASU, US). urn:lsid:ipni.org:names:77134879-1
Name of uncertain application
Myriopteris cheiloglyphisFée, Mém. Fam. Foug. 8: 77. 1857.
Excluded names
Myriopteris contracta(Kunze) Fée, Mém. Fam. Foug. 5: 149. 1852. = Cheilanthes contracta (Kunze) Mett. ex Kuhn
Myriopteris hirta(Sw.) J. Sm., Ferns Brit. and For. 174. 1866. = Cheilanthes hirta Sw.
Myriopteris induta (Kunze) Fée, Mém. Fam. Foug. 5: 149. 1852. = Cheilanthes induta Kunze
Myriopteris intermedia(Kunze) Fée, Mém. Fam. Foug. 5: 149. 1852. = Cheilanthes hirta Sw. fide
Myriopteris macleaniiJ. Sm., Hist. Fil. 280. 1875. = Cheilanthes pilosa Goldm. fide
Myriopteris scariosa(Sw.) Fée, Mém. Fam. Foug. 5: 149, t. 29, f. 6. 1852. = Cheilanthes scariosa Sw.
Myriopteris szovitzii(Fisch. & Meyer) J. Sm., Hist. Fil. 281. 1875. = Cheilanthes persica (Bory) Mett. ex Kuhn fide
The authors thank A. R. Smith, R. Moran, K. N. Gandhi, and L. J. Dorr for assistance interpreting and/or obtaining obscure taxonomic literature. We also extend our appreciation to K. M. Pryer, G. J. Gastony, G. Yatskievych, L. Huiet, E. M. Sigel, F.-W. Li, and C. J. Rothfels for helpful comments on the manuscript. This study was completed in partial fulfillment of a doctoral dissertation in Biology at Duke University by the first author. Research support to A. L. G. was provided by a Society for Systematic Biologists Graduate Student Research Award, the American Society of Plant Taxonomists Shirley and Alan Graham Graduate Student Research Grant, as well as an NSF Doctoral Dissertation Improvement Grant (NSF-DDIG 1110767). Additional funding for this project was provided by NSF-DEB 0717398 awarded to M.D.W.